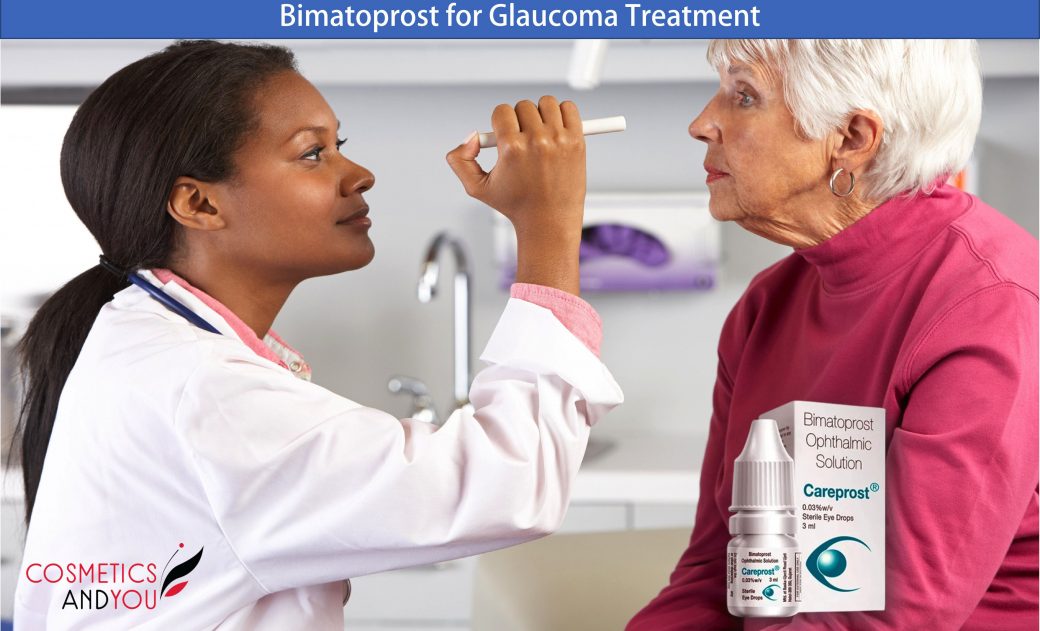
Bimatoprost is a prostamide that has been proven highly effective as monotherapy, adjunctive, and replacement therapy for lowering intraocular pressure and providing good control of intraocular pressure (IOP) in patients with open-angle glaucoma and ocular hypertension.
Bimatoprost for the treatment of glaucoma
Ophthalmic topical bimatoprost is a prostaglandin that has received approval from the United States Food and Drug Administration (FDA) as a treatment for lowering intraocular pressure (IOP). Evidence suggests that topical bimatoprost is among the most effective medicine for reducing IOP in eyes, ocular hypertension, and primary open-angle glaucoma. Glaucoma is an eye disorder with the chronic progression of visual field defects because of the loss of retinal ganglion cells. Glaucoma treatment aims to control the progression of visual field defects. Research studies indicate that control of visual field defects is associated with a decrease in IOP. Typically, a decrease in the IOP is achieved by administering single-drug eye drops.
Growth of eyelashes is a well-documented side effect of bimatoprost during the treatment of glaucoma. Later the FDA approved ophthalmic bimatoprost to treat hypotrichosis of eyelashes, such as for patients post-chemotherapy. Because of longer, darker, and thicker eyelashes, bimatoprost 0.03% ophthalmic solution is often purchased for cosmetic purposes. The ophthalmic product is available at 0.03% in the United States when used to lower IOP; daily dosing of bimatoprost may be used alone or with another drug.
Mechanism of action in reduction of intraocular pressure
It has been well documented that bimatoprost and several topical prostaglandin analogues sustainably and effectively reduce IOP and are used as a standard first-line treatment of open-angle glaucoma. This is achieved primarily through enhancing aqueous through the uveoscleral pathway. It is shown that lowering IOP in glaucoma and ocular hypertension patients may reduce the risk of disease progression.
Applying bimatoprost ophthalmic solution of open-angle glaucoma
Bimatoprost ophthalmic solution 0.03% may be applied directly to the ocular surface as an eye drop. The usual dose of bimatoprost is one drop every evening, although it may vary depending on the ophthalmologist’s dosing. A patient should wait five minutes between administering bimatoprost and other ophthalmic drugs. With bimatoprost, a 0.03% reduction of IOP should begin after four hours, and the effect will peak at twelve hours but will maintain the reduced IOP through 24 hours. Research reports of one year demonstrate that bimatoprost effectively lowers IOP long-term and is generally well tolerated.
Side effects
Numerous clinical trials have demonstrated the high levels of safety and tolerance of bimatoprost when applied to the ocular surface. Several studies of bimatoprost eye drop application revealed that other than transient eyelash prominence, the most common side effects included eye pruritus, conjunctival hyperaemia, burning irritation, dry eye syndrome, eye pain, visual disturbances, foreign body sensations, and pigmentation of the eyelid margin. Use of this ophthalmic agent may not be appropriate for some people with closed-angle glaucoma and inflammatory or neovascular glaucoma. Contact lenses should be taken off before the application of this ophthalmic solution. Lenses can be reinserted after fifteen minutes.
Contraindications
Bimatoprost is contraindicated in people with a medical history of bimatoprost hypersensitivity or hypersensitivity to other Intraocular applicators containing Bimatoprost eye drops for glaucoma is contraindicated in infants and children due to concern for pigmentation changes with long term usage. The safety and effectiveness of bimatoprost application for hypotrichosis have not been studied in children and infants younger than five. The usage should not be considered if the potential benefits justify the risks. This careprost ophthalmic agent has not been adequately studied in pregnant women.
Summary: Patients on bimatoprost therapy achieve low IOP levels that are maintained throughout the day and night, and long-term trials have shown that the effectiveness of bimatoprost is sustained. The ophthalmic agent has been proven to be safe and well tolerated in clinical trials and, as a once-daily drug, allows for patient convenience resulting in better treatment compliance. It is the most effective ophthalmic solution for protecting the visual field in patients with ocular hypertension and glaucoma. you can buy careprost online from cosmeticsandyou at affordable prices.